Breathless doing nothing. Unable to lie flat. What might be the reason?
A paralysed or weakened diaphragm can reduce lung capacity by 20–30% on its own. If you've been told your heart and lungs are fine but you're still struggling to breathe — the diaphragm may be the explanation no one has looked for yet.

For some patients, it started after a cardiac operation — a bypass, a valve repair, an ablation procedure. The surgery went well. But recovering from it was harder than expected, the breathlessness lingered, and every follow-up appointment produced the same reassurance: "your heart is fine." They weren't wrong. But the phrenic nerve — the nerve that controls your diaphragm — was damaged during the procedure. Nobody told you. And nobody looked.
For others, it arrived differently — after a car accident, a neck injury, a virus, or seemingly from nowhere. The diaphragm on one side no longer drops when you breathe in. Instead it rises — compressing your lung from below, stealing 20–30% of your breathing capacity every single breath. You can't climb stairs without stopping. You can't lie down to sleep. The world has quietly contracted around the limitations of your breath.
"The tragedy of diaphragm paralysis is not the condition itself — it is how long patients live with it before anyone identifies what's actually wrong. A single chest X-ray showing an elevated hemidiaphragm is often the first clue that has been sitting in someone's notes for years, unrecognised. Once we see it, there is almost always something we can do."
Diaphragmatic plication is a surgical procedure that folds and secures the paralysed portion of the diaphragm, restoring the normal geometry of the chest and dramatically improving lung function. In the right patient, 80–95% experience meaningful improvement in breathlessness — often dramatic. This guide explains everything: what the diaphragm does, what goes wrong, how it's diagnosed, and what surgery involves.
Seen within days. No referral needed. Bring any existing chest X-rays, CT scans, or lung function results — no need to repeat investigations unnecessarily.
"I had a CABG three years ago and never got my breath back properly. Everyone said my heart was fine. I slept in a recliner for two and a half years. Mr Scarci was the first doctor who looked at my diaphragm. VATS plication was done on a Thursday. I was home on Friday. I slept flat in my own bed for the first time in years by Saturday night. I cried."
Diaphragmatic plication is a technically demanding procedure performed by a small number of thoracic surgeons. Experience, operative volume, and dedicated expertise make a measurable difference to outcomes.
Mr Scarci has published his own minimally invasive VATS plication technique. This is not a procedure he performs occasionally — it is a core part of his specialist practice, and his results reflect that experience.
Most patients are discharged the day after VATS plication. This is only possible with a minimally invasive technique performed by a surgeon with extensive experience. It is not the norm elsewhere for this procedure.
Many patients arrive having been told their breathlessness is cardiac, respiratory, or anxiety-related — with an elevated diaphragm sitting in the imaging notes unrecognised. Mr Scarci investigates the diaphragm specifically.
You speak directly with Mr Scarci. Post-operative concerns are addressed promptly and personally. For a procedure where breathing is the outcome, close follow-up is not optional — it is part of the care.
Most patients are seen within one week of contact. When breathlessness is significantly impairing daily life and sleep, waiting months on a waiting list is not the right answer.
All major UK private insurers are accepted. Diaphragmatic plication is covered as a medically necessary procedure. Transparent self-pay pricing is also available with no hidden extras.
The diaphragm is a large dome-shaped muscle sitting at the base of your chest, separating the chest cavity from the abdomen. When you inhale, it contracts and flattens downward — expanding the chest cavity and drawing air into the lungs. When you exhale, it relaxes and rises. This happens around 20,000 times each day, driven by signals from the phrenic nerve, which originates in the neck at cervical levels C3, C4, and C5.
The diaphragm accounts for approximately 70–80% of breathing effort at rest. When one side is paralysed, instead of moving downward on inhalation, it moves paradoxically upward — compressing the lung beneath it and working directly against breathing. The affected lung cannot expand fully. Lung capacity on that side may be reduced by 20–30%.
This is why diaphragm paralysis causes not just exertional breathlessness but profound orthopnoea — inability to lie flat. When you lie down, abdominal contents press against the paralysed diaphragm from below, worsening the compression. Patients sleep semi-reclined, in chairs, or with multiple pillows to avoid this — sometimes for years before anyone identifies the cause.
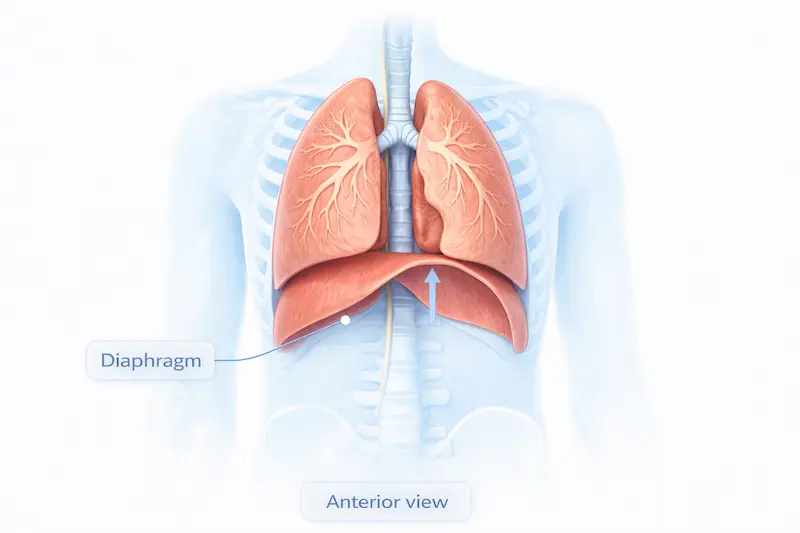
The phrenic nerve is the fragile thread that controls your diaphragm. Anything that damages it — surgery, trauma, compression, or infection — can interrupt the signal and paralyse the muscle it controls.
Phrenic nerve injury is a recognised complication of cardiac bypass surgery, valve procedures, and AF ablation. The nerve passes close to the heart and can be stretched, cooled, cauterised, or cut. Post-surgical diaphragm paralysis is underdiagnosed — patients are often told their breathlessness is "post-operative" or cardiac, and the diaphragm is never investigated.
Lobectomy, pneumonectomy, and mediastinal operations can injure the phrenic nerve either directly or through thermal injury. VATS carries lower risk than open surgery, but phrenic nerve injury remains possible, particularly for complex mediastinal cases.
An idiopathic inflammatory condition causing sudden severe shoulder pain followed by weakness and paralysis — including of the phrenic nerve. Recovery is possible but slow; diaphragm paralysis may persist for years after the initial episode.
Road traffic accidents, falls, or direct blows to the neck or chest can stretch or tear the phrenic nerve. The cervical spine injury may have been recognised and treated, while the resulting diaphragm paralysis goes unnoticed for months or years.
Mediastinal tumours, enlarged lymph nodes, lung cancer, or thyroid masses can compress the phrenic nerve. Breathlessness in a patient with known cancer that develops progressively should prompt evaluation of diaphragm function.
In up to 40–50% of cases, no clear cause can be identified. This does not mean treatment is unavailable — surgery remains equally effective regardless of whether the cause is known.
Some individuals are born with a thin, poorly muscularised hemidiaphragm that bulges upward into the chest. This may be asymptomatic in childhood but become progressively problematic in adulthood. Plication is highly effective for eventration.
Shingles affecting the cervical dermatomes, viral neuritis, and COVID-19-associated nerve injury have all been reported as causes. Post-COVID breathlessness that doesn't improve should prompt diaphragm evaluation.
Phrenic nerve injury is one of the most under-recognised complications of cardiac procedures. Studies suggest it occurs in up to 20–30% of cases involving hypothermic cardioplegia and significantly in AF ablation procedures. In many patients, it is never identified because breathlessness post-cardiac surgery has many possible explanations, and the diaphragm is rarely investigated.
If you had cardiac surgery — bypass, valve repair, TAVI, or AF ablation — and your breathlessness never fully resolved, your diaphragm should be formally assessed. A fluoroscopy (sniff test) takes minutes, is entirely non-invasive, and provides a definitive answer. Plication surgery, when indicated, typically produces dramatic improvement even in patients who have been struggling for years.
The symptoms of diaphragm paralysis are dominated by breathlessness — but the specific pattern is distinctive and, once recognised, highly characteristic.
Disproportionate breathlessness with activities that used to be effortless — climbing stairs, walking uphill, carrying shopping. Often attributed to deconditioning or cardiac causes.
Breathlessness that dramatically worsens lying down is the most characteristic symptom of diaphragm paralysis. Patients sleep reclined, in chairs, or propped on multiple pillows. Often the single most life-disrupting symptom.
Waking breathless, frequent nocturnal arousal, non-restorative sleep, and morning headache from overnight hypoventilation. May mimic or co-exist with sleep apnoea.
The paralysed, elevated diaphragm impairs mucus clearance from the lower lung on the affected side. Repeated pneumonia or bronchitis on the same side should raise suspicion.
The additional effort of compensatory breathing depletes energy. Many patients describe a profound, disproportionate fatigue that makes ordinary daily activities exhausting.
A full stomach pushes the abdominal contents upward, further elevating the paralysed diaphragm. Breathlessness that reliably worsens after eating is a clue to diaphragmatic cause.
The investigation of diaphragm paralysis uses a specific set of tests — most of which are not routinely ordered in a standard breathlessness workup. The key tests are straightforward once the diagnosis is considered.
The classic finding on a plain chest X-ray is an elevated hemidiaphragm — typically 3–5 cm higher than the normal side. This is often described in X-ray reports as an incidental finding and then ignored. An elevated hemidiaphragm in a breathless patient should always prompt formal diaphragm assessment.
Real-time X-ray (fluoroscopy) of the diaphragm during a sharp sniff inhalation. In a normal diaphragm, both sides move downward simultaneously. In paralysis, the affected side moves paradoxically upward — confirming the diagnosis. The sniff test is the gold standard for diagnosing unilateral diaphragm paralysis and takes minutes to perform.
Real-time ultrasound can visualise diaphragm movement and measure its thickness during contraction. Useful as an accessible, radiation-free alternative to fluoroscopy, and particularly helpful for assessing bilateral paralysis. An experienced sonographer performing a dedicated diaphragm assessment is needed — not a standard abdominal ultrasound.
Spirometry in both the upright and supine positions is a simple and revealing test. In diaphragm paralysis, the FVC (forced vital capacity) drops by more than 25–30% when lying down — this positional difference is highly characteristic and clinically significant. Standard spirometry performed only when upright may appear near-normal.
Electrophysiological assessment of phrenic nerve function — measuring conduction velocity and amplitude. Confirms the diagnosis and helps determine whether the nerve is entirely absent (complete palsy), markedly reduced (severe paresis), or partially preserved. Guides decisions about whether phrenic nerve reconstruction might be appropriate alongside or instead of plication.
Identifies any structural cause of phrenic nerve compression — mediastinal mass, enlarged lymph nodes, apical lung tumour, or thyroid pathology. Essential before any surgical planning for plication, and to exclude a remediable compressive cause that should be treated first.
The word "plication" comes from the Latin plicare — to fold. In diaphragmatic plication, the surgeon folds and sutures the weakened or paralysed portion of the diaphragm, creating a tightened, tensioned surface that sits at a more normal position in the chest.
The procedure does not restore nerve function or cause the diaphragm to contract actively again. What it does is eliminate the paradoxical upward movement that is working against every breath. By creating a fixed, tensioned platform, it allows the remaining functional lung on that side to expand more fully, and prevents the abdominal contents from pushing the diaphragm into the chest.
The result: more lung space, less paradoxical movement, better lung mechanics — and for most patients, a dramatic improvement in breathlessness, sleep quality, and exercise tolerance.
The surgical approach is tailored to the patient — their anatomy, fitness, and the complexity of the case. Minimally invasive techniques are the preferred approach for most patients. The goal of all approaches is identical: to fold the diaphragm upon itself using a series of sutures, creating a taut, flat platform that sits at a lower, more normal position.
Video-Assisted Thoracoscopic Surgery uses small incisions and a high-definition camera to access the diaphragm through the chest. The diaphragm is folded and secured with sutures under direct vision. Mr Scarci has published his own VATS plication technique — patients typically go home the following day with minimal pain, and the recovery is dramatically faster than with open surgery.
Robotic surgery offers enhanced visualisation and instrument articulation through similarly small incisions — particularly useful for complex cases where precise suture placement is critical, or where obesity or previous surgery makes thoracoscopic work more technically demanding.
The traditional approach, requiring a larger incision through the chest wall. Occasionally still necessary for very complex cases, bilateral plication, or when other procedures need to be performed at the same time. Recovery is longer — typically 4–7 days in hospital.
Plication performed through the abdomen rather than the chest — using laparoscopic instruments to fold the diaphragm from below. An option when the thoracic route is contraindicated (previous thoracic surgery with adhesions, for example).
Most patients are surprised by how quickly they feel the benefit — and how manageable the recovery is with a minimally invasive approach.
VATS plication is performed under general anaesthetic, typically taking 1–2 hours. A chest drain is left briefly to remove any air or fluid. Pain is well-managed. Most VATS patients go home the following day — a striking contrast with the traditional open approach, which required 5–7 nights in hospital.
Mild discomfort at the port sites. Breathing exercises — particularly incentive spirometry — are important to maximise the benefit of the newly restored diaphragm position. Most patients notice improvement in breathlessness remarkably quickly after surgery, often within the first 1–2 weeks.
Gradual return to normal activities. Driving and return to desk work typically in weeks 3–4. Pulmonary rehabilitation — supervised aerobic exercise — maximises the functional gain from restored lung expansion and helps rebuild the conditioning lost during months or years of breathlessness.
Repeat pulmonary function testing typically shows measurable improvement in FVC and exercise capacity. Orthopnoea usually resolves completely — patients can lie flat for the first time in months or years. Energy levels improve. Many patients describe this phase as feeling better than they have in years.
Published data show that the majority of patients maintain their breathing improvement for many years after plication. Regular follow-up with chest X-ray and spirometry monitors the durability of the result. In the small minority where plication fails or recurrence occurs, revision surgery is possible.
Plication is the most effective intervention for most patients with symptomatic diaphragm paralysis — but it is not the only option. The right treatment depends on the cause, severity, and whether the phrenic nerve has any preserved function.
For mild or recently acquired unilateral paralysis, watchful waiting is appropriate — some cases (particularly post-viral or traumatic) spontaneously recover over months. Regular review with pulmonary function tests and symptomatic assessment is needed. Surgery is reserved for patients with persistent significant symptoms beyond 12–18 months.
CPAP or BiPAP overnight can support breathing during sleep in bilateral paralysis or in patients not fit for surgery. Manages symptoms but does not address the underlying mechanical problem. May be appropriate as a bridge to surgery or for patients unsuitable for an operation.
An implanted device stimulates the phrenic nerve electrically to contract the diaphragm. Requires a functioning nerve — only appropriate if phrenic nerve conduction studies confirm preserved nerve viability. Not widely available; specialist centres only.
Surgical repair or reconstruction of the phrenic nerve — decompression, nerve grafting, or neurotisation — aims to restore active diaphragm function. Best suited to selected cases of relatively recent phrenic nerve injury where the diaphragm muscle has not atrophied irreversibly.
If a paralysed or weakened diaphragm is the explanation for your breathlessness, diaphragmatic plication can change your life. A specialist consultation determines whether you are a candidate — and gives you a clear plan for what happens next.